Public Health Agency on X: "If your planning on going to university or college make sure your teenage immunisations are up-to-date. It's also worth checking if you got your MMR and HPV.

Antibody persistence and booster response following MenACWY-CRM vaccination in children as assessed by two different assay methods - ScienceDirect
![MENVEO (Meningococcal [Groups A,C,Y and W-135] Oligosaccharide Diphtheria CRM197 Conjugate Vaccine) | Home MENVEO (Meningococcal [Groups A,C,Y and W-135] Oligosaccharide Diphtheria CRM197 Conjugate Vaccine) | Home](https://gskpro.com/content/dam/global/hcpportal/en_US/img/menveo/hero-mobile-image.png)
MENVEO (Meningococcal [Groups A,C,Y and W-135] Oligosaccharide Diphtheria CRM197 Conjugate Vaccine) | Home

Full article: Immunogenicity and safety of MenACWY-TT, a quadrivalent meningococcal tetanus toxoid conjugate vaccine recently licensed in the United States for individuals ≥2 years of age

NHS North Central London on X: "The MenACWY vaccine is routinely given in school years 9-10, at the same time as the 3-in-1 booster. Together they protect against tetanus, diphtheria, polio, meningitis,
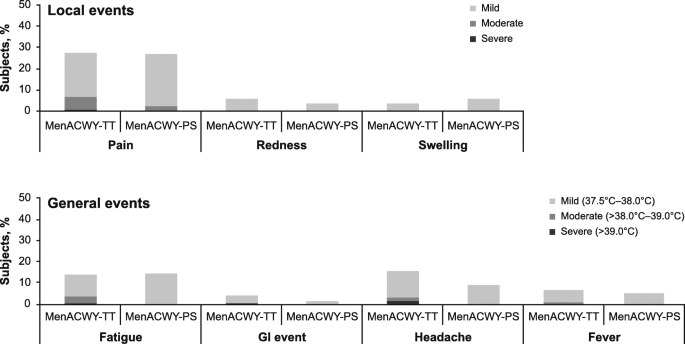
A phase 2b/3b MenACWY-TT study of long-term antibody persistence after primary vaccination and immunogenicity and safety of a booster dose in individuals aged 11 through 55 years | BMC Infectious Diseases
![Meningococcal Conjugate Vaccine | Menactra® (Meningococcal [Groups A, C, Y, and W-135] Polysaccharide Diphtheria Toxoid Conjugate Vaccine) Meningococcal Conjugate Vaccine | Menactra® (Meningococcal [Groups A, C, Y, and W-135] Polysaccharide Diphtheria Toxoid Conjugate Vaccine)](https://www.menactra.com/_assets/img/menactra-web-timeline-art-mobile.jpg)
Meningococcal Conjugate Vaccine | Menactra® (Meningococcal [Groups A, C, Y, and W-135] Polysaccharide Diphtheria Toxoid Conjugate Vaccine)
Immunogenicity Booster | MENVEO [Meningococcal (Groups A, C, Y and W-135) Oligosaccharide Diphtheria CRM₁₉₇ Conjugate Vaccine]

Notes from the Field: Increase in Meningococcal Disease Among Persons with HIV — United States, 2022 | MMWR

MenACWY-CRM conjugate vaccine booster dose given 4–6 years after priming: Results from a phase IIIb, multicenter, open label study in adolescents and adults - ScienceDirect
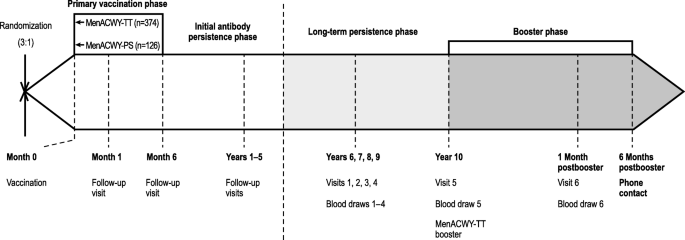
A phase 2b/3b MenACWY-TT study of long-term antibody persistence after primary vaccination and immunogenicity and safety of a booster dose in individuals aged 11 through 55 years | BMC Infectious Diseases


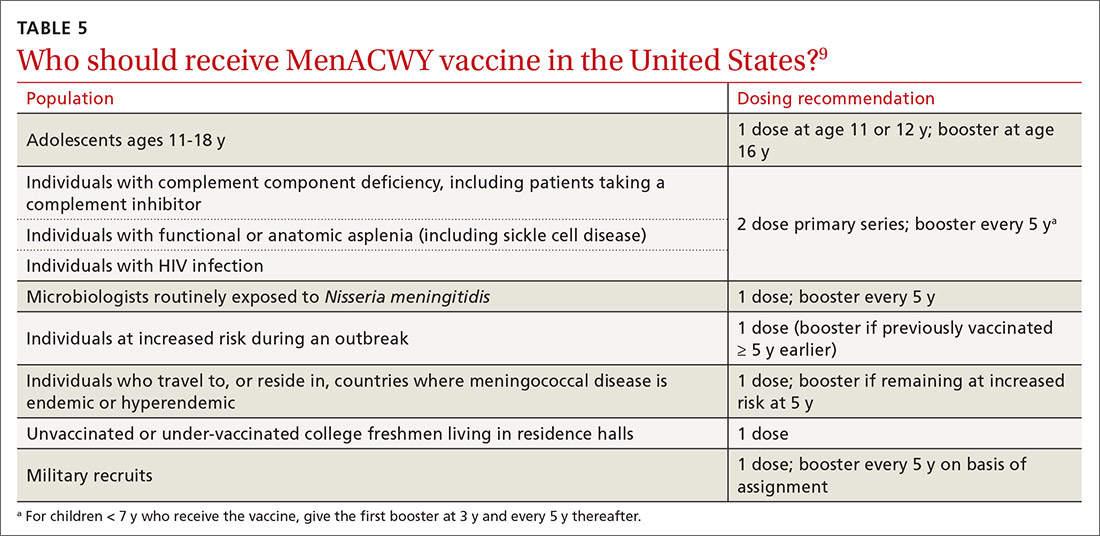

:max_bytes(150000):strip_icc()/Meningococcal-vaccine-5215620_final_05-063b0877ce5c401ca3a0545a27ca6e3c.jpg)