FDA vaccine advisers 'disappointed' and 'angry' that early data about new Covid-19 booster shot wasn't presented for review last year | CNN
Pfizer asks FDA to authorize Covid booster shots that target omicron BA.5 for people ages 12 and older

ATAGI recommendations on use of the Pfizer bivalent (Original/Omicron BA.1) COVID-19 vaccine | Australian Government Department of Health and Aged Care
Common Questions About Bivalent COVID-19 Boosters | Johns Hopkins | Bloomberg School of Public Health

BioNTech SE on X: "We completed, together with @Pfizer, the submission to the @US_FDA requesting Emergency Use Authorization (EUA) of a 10-µg booster dose of the Omicron BA.4/BA.5-adapted bivalent COVID-19 vaccine for

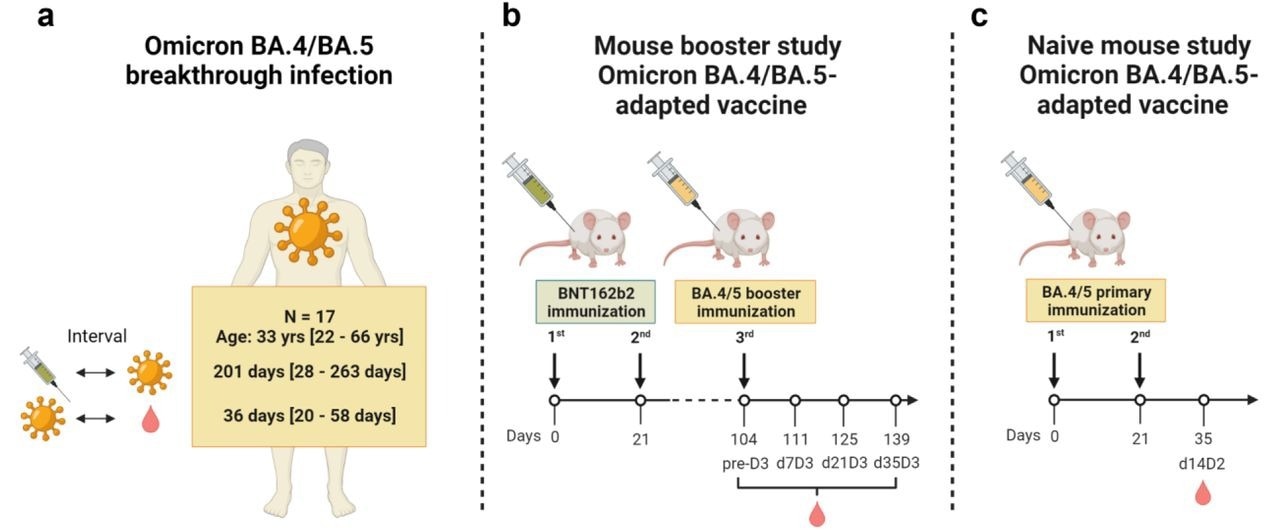
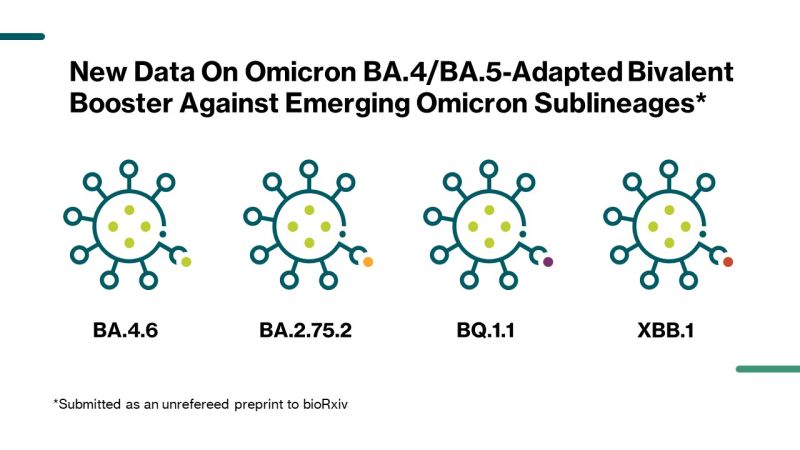
Low neutralization of SARS-CoV-2 Omicron BA.2.75.2, BQ.1.1 and XBB.1 by parental mRNA vaccine or a BA.5 bivalent booster | Nature Medicine


/cloudfront-us-east-2.images.arcpublishing.com/reuters/I6XWR6AYYFLQ3NGU2WCG67F7IY.jpg)