ATAGI statement on use of the Moderna bivalent Original/Omicron vaccine | Australian Government Department of Health and Aged Care

Omicron Subvariant Now Dominant in U.S.; FDA, CDC OK Second Booster for Adults Over 50 | The Hospital of Central Connecticut | CT
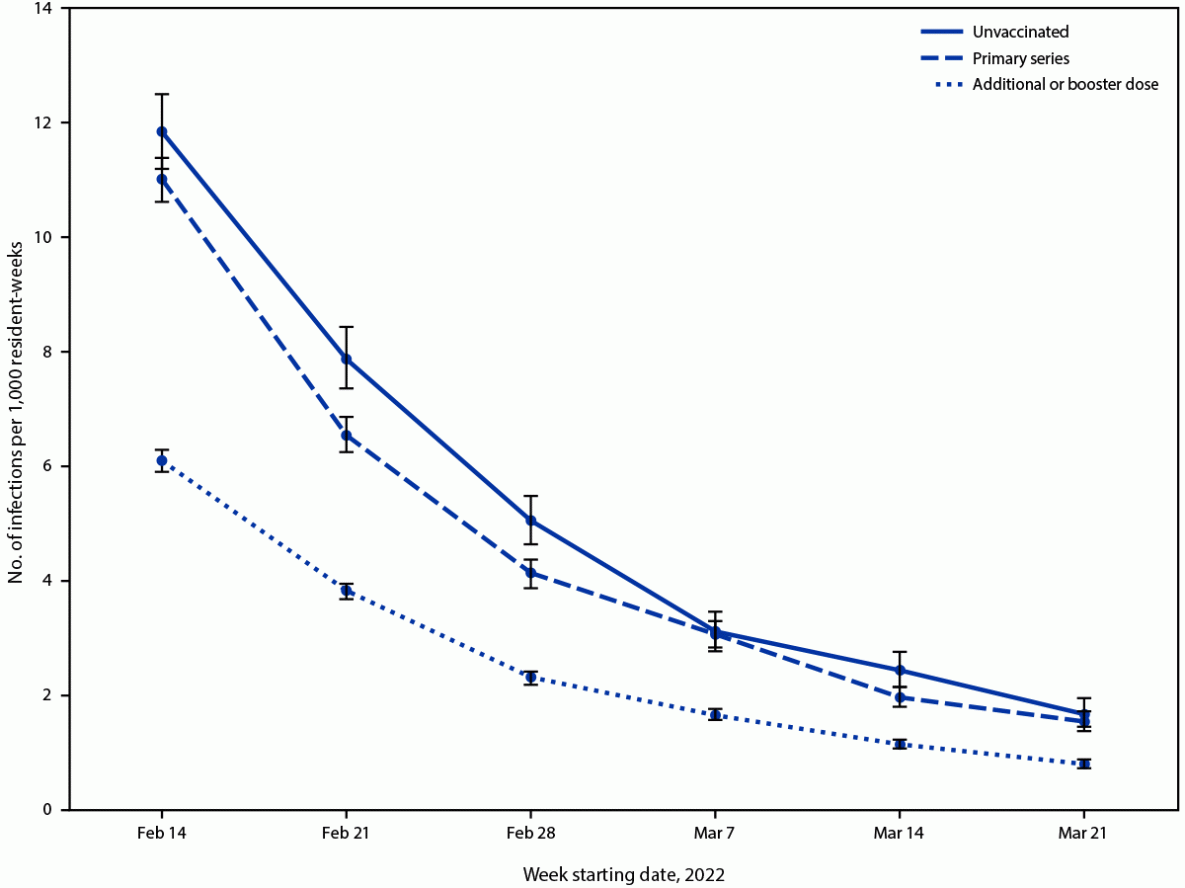
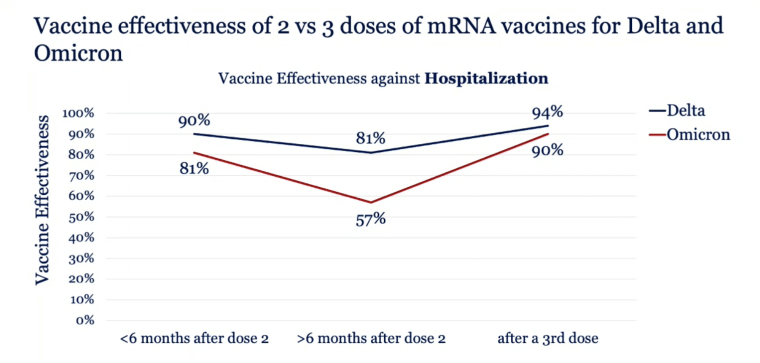
Effectiveness of a COVID-19 Additional Primary or Booster Vaccine Dose in Preventing SARS-CoV-2 Infection Among Nursing Home Residents During Widespread Circulation of the Omicron Variant — United States, February 14–March 27, 2022 | MMWR
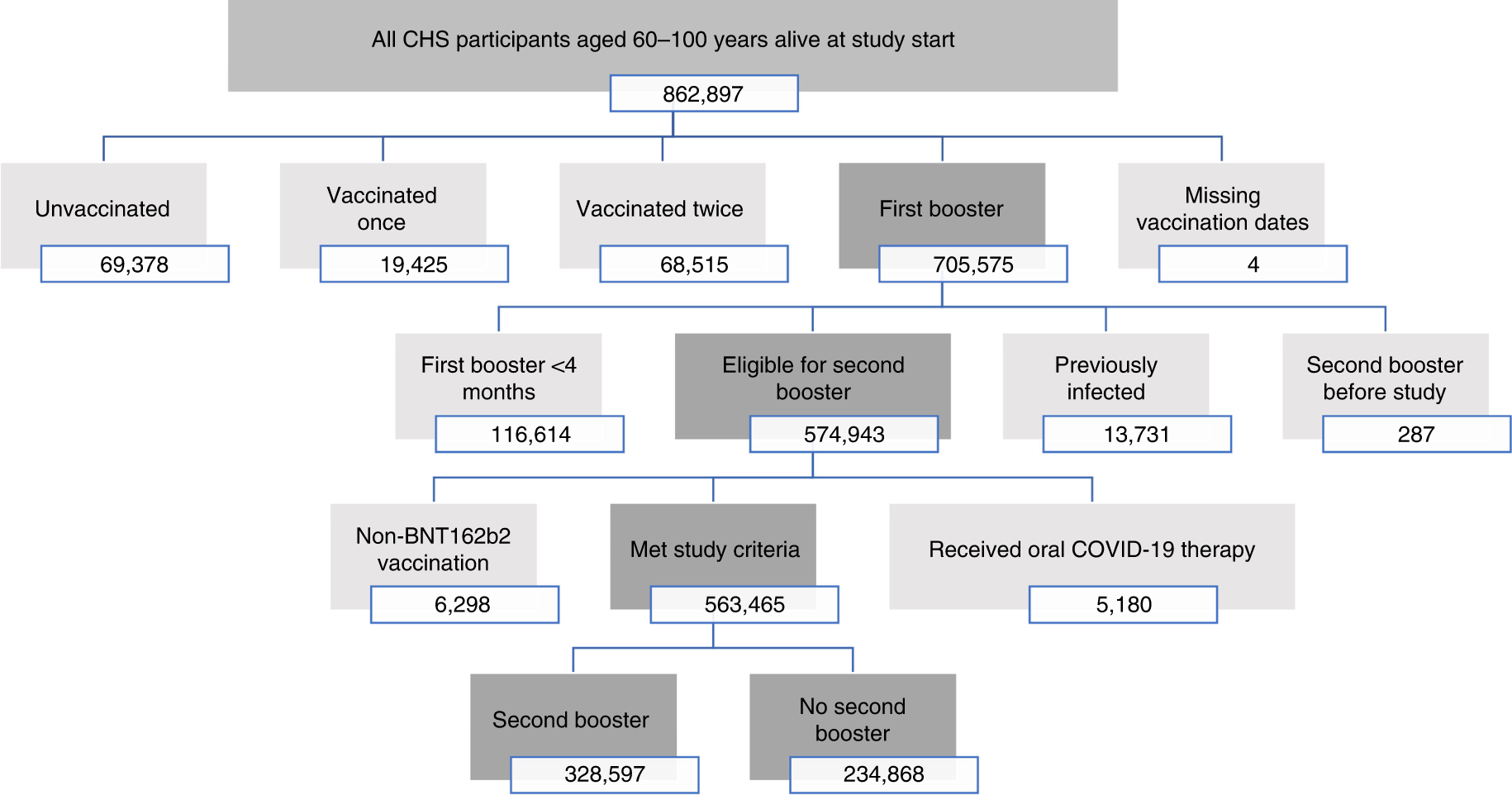
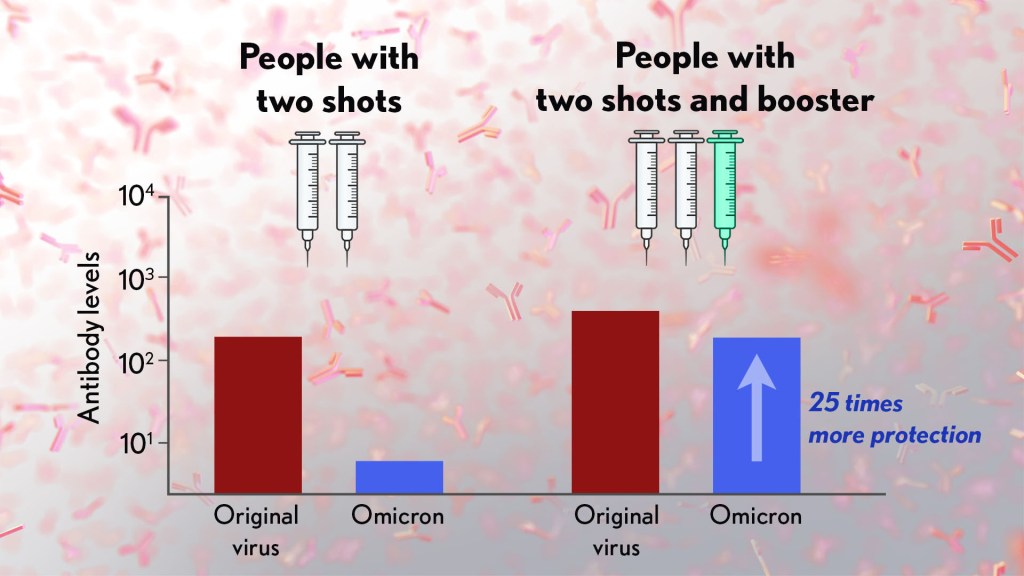
Effectiveness of a second BNT162b2 booster vaccine against hospitalization and death from COVID-19 in adults aged over 60 years | Nature Medicine

Severity of omicron variant of concern and effectiveness of vaccine boosters against symptomatic disease in Scotland (EAVE II): a national cohort study with nested test-negative design - The Lancet Infectious Diseases

Effectiveness of Bivalent mRNA Vaccines in Preventing Symptomatic SARS-CoV-2 Infection — Increasing Community Access to Testing Program, United States, September–November 2022 | MMWR
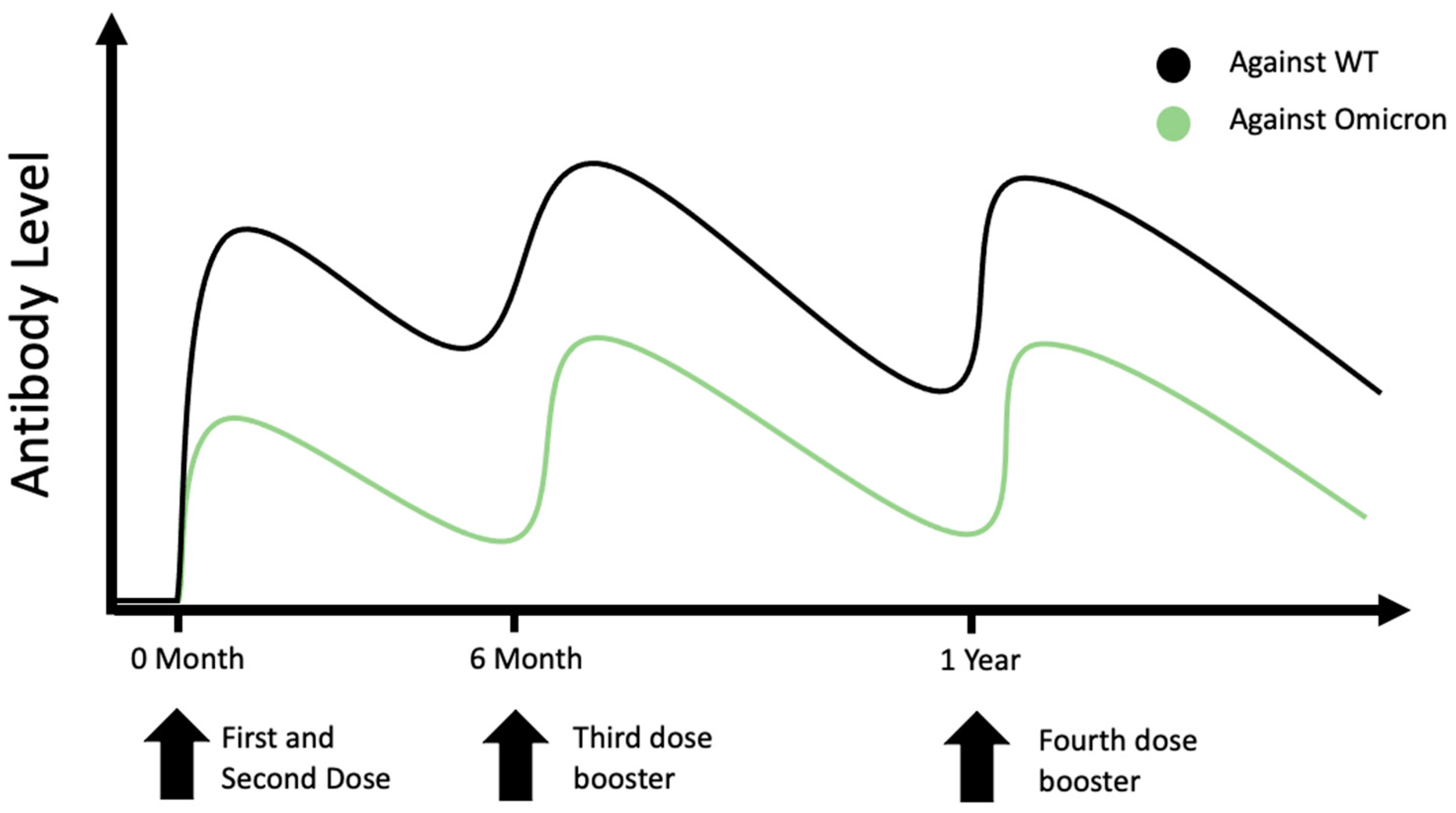
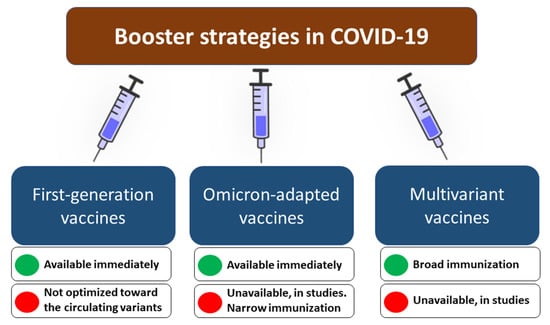
Quarta dose contro Covid-19: perché, a chi, quando e con quale vaccino - Network Bibliotecario Sanitario Toscano

Second Pfizer/BioNTech bivalent COVID-19 booster vaccine approved by UK medicines regulator - GOV.UK